 2010 Dietary Guidelines is a series in which I discuss the Dietary Guidelines for Americans, 2010 released by the UDSA and US Department of Health and Human Services at the end of February. In the series, I attempt to untangle the facts from the political rhetoric and scientific jargon, to translate the key data & recommendations into plain English, and to investigate whether the document truly reflects the best and most current food science available today.
2010 Dietary Guidelines is a series in which I discuss the Dietary Guidelines for Americans, 2010 released by the UDSA and US Department of Health and Human Services at the end of February. In the series, I attempt to untangle the facts from the political rhetoric and scientific jargon, to translate the key data & recommendations into plain English, and to investigate whether the document truly reflects the best and most current food science available today.
Need to catch up?
—————————————————————-
Fat 102: Hydrogenation, Trans fats and Essential Fatty Acids, Oh My!
Hello, kids, and welcome back! Please open your textbooks and turn to page 394. OK, I realize that you don’t have textbooks, and we’re not actually going to talk about werewolves today (and I’m not quite as cranky as Professor Snape), but we are going to talk about hydrogenation and trans fats: almost as scary. Before we get down to the nitty gritty, however, let’s do a brief recap of what we learned last week in Fat 101:
- dietary fat molecules are triglycerides, consisting of 3 fatty acid chains bound at one end to a glycerol
- fatty acids can be saturated (every free carbon is bound to a hyrdrogen) or unsaturated (some free carbon sites are bound to a neighboring carbon, resulting in a carbon double bond)
- saturated fatty acids lay flat, while unsaturated fatty acids have kinks in their structure caused by the carbon double bonds
- unsaturated fatty acids can be monounsaturated (one carbon double bond) or polyunsaturated (two or more double bonds); the number of double bonds affects the structure (number of kinks) of the fatty acid
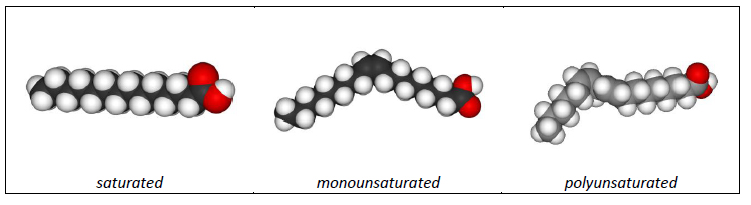
- saturated fats are solid at room temperature and typically come from animal sources; unsaturated fats are liquid at room temperature and come from vegetable sources; all dietary fats contain a combination of saturated, mono- and polyunsaturated fatty acids
HYDROGENATION
While we did a boatload of learning on the nature of fat last week, we have to delve further into the chemistry in order to understand fats as our food manufacturers use them. To kick off today’s class, we are going to talk about hydrogenation. Recall that the white balls in the above molecular models represent hydrogen atoms: hydrogenation is simply the process of adding hydrogen to fatty acids. Since saturated fats already have a full complement of hydrogen, hydrogenation can only be performed on unsaturated fats, whereupon one or more of the carbon double bonds on the fatty acid is broken and replaced with a hydrogen bond (warning – more chemistry ahead!).
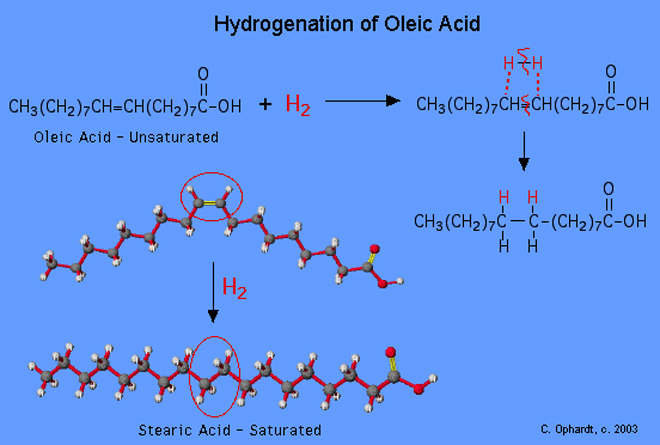 Consider the diagram above (ignore the chemical formulas for the moment and just focus on the molecular model at bottom left): the top molecular model represents oleic acid, the primary fatty acid in olive oil (“oleic” meaning ” of olive“). You can see that oleic acid is an unsaturated fatty acid by its kinked structure; in fact, it is a monounsaturated fatty acid, with a single carbon double bond in the middle of the chain (circled in red). The addition of two hydrogen molecules to oleic acid eliminates the carbon double bond and produces stearic acid, a naturally-occuring saturated fatty acid that is a common component of lard.
Consider the diagram above (ignore the chemical formulas for the moment and just focus on the molecular model at bottom left): the top molecular model represents oleic acid, the primary fatty acid in olive oil (“oleic” meaning ” of olive“). You can see that oleic acid is an unsaturated fatty acid by its kinked structure; in fact, it is a monounsaturated fatty acid, with a single carbon double bond in the middle of the chain (circled in red). The addition of two hydrogen molecules to oleic acid eliminates the carbon double bond and produces stearic acid, a naturally-occuring saturated fatty acid that is a common component of lard.
So, you can see where the chemistry is fascinating, no? These two fatty acids are identical in every respect but one: both have 18 carbon atoms, both have the same carboxyl group (the red balls on the right hand side) and both can bind to the same glycerol; the addition of just two hydrogen atoms (or water, H2O, minus the oxygen) changes olive oil to lard. (I know I’m a geek, but these things never fail to amaze me).
 How, exactly, do we add hydrogen to unsaturated fats to form hydrogenated fats? Well, it’s not as simple as putting unsaturated fats and hydrogen in the same room (a little soft lighting, a little Marvin Gaye). The process of hydrogenation has been around since the turn of the century and is credited to Paul Sabatier, a French chemist (who was awarded the Nobel Prize in 1912 for his work on the subject). Essentially, hydrogen gas is bubbled through vegetable oil (unsaturated fat) in the presence of a catalyst, usually nickel or another metal. The catalyst, like a professional matchmaker with shy singletons, facilitates the breaking of the carbon double bond in the unsaturated fat and the subsequent binding of hydrogen to the free carbon.
How, exactly, do we add hydrogen to unsaturated fats to form hydrogenated fats? Well, it’s not as simple as putting unsaturated fats and hydrogen in the same room (a little soft lighting, a little Marvin Gaye). The process of hydrogenation has been around since the turn of the century and is credited to Paul Sabatier, a French chemist (who was awarded the Nobel Prize in 1912 for his work on the subject). Essentially, hydrogen gas is bubbled through vegetable oil (unsaturated fat) in the presence of a catalyst, usually nickel or another metal. The catalyst, like a professional matchmaker with shy singletons, facilitates the breaking of the carbon double bond in the unsaturated fat and the subsequent binding of hydrogen to the free carbon.
If you recall our analogy of the line of pre-school children from the last post, linked hand in hand and wearing white hats & shoes , think about the children representing an unsaturated fat: two kids in the middle of the chain take off their hats and lean their heads together (whispering, giggling), representing the carbon double bond. The teacher comes along, tells the kids to stand up straight and put on their hats. The teacher is the metal catalyst, facilitating the reaction; the hats are the hydrogen atoms, now firmly “bonded” to the kids’ heads. The kinked line of children, caused by the two whispering renegades in the middle, is now straight, and “saturated” with hats, a.k.a. hydrogen.
The oleic acid example is a simple one of processing a monounsaturated fat into a saturated fat, or hydrogenated vegetable oil, by adding 2 hyrdrogen atoms (you can see the deception in food labeling here; “hydrogenated fat” would seem to imply a solid, like Crisco; “hydrogenated vegetable oil” which means exactly the same thing, seems to imply a liquid, and will be perceived as “healthier” by the consumer). In practice, food manufacturers use polyunsaturated vegetable oils, like soybean, canola and cottonseed oil, to create partially hydrogenated vegetable oil, used in a myriad of manufactured foods, including crackers & chips, cereals & granola bars, baking mixes, cookies & cakes of all kinds. Varying the conditions under which the vegetable oil is hydrogenated, including temperature, pressure, and amount of hydrogen & catalyst, yields differing amounts of hydrogenated fatty acids and results in an end product with slightly different properties; texture, “plasticity” (spreadability), shelf-life.
 So hydrogenation is the process of transforming a liquid oil to a solid or semi-solid fat by breaking carbon double bonds and replacing them with hydrogen-saturated single bonds. Although the reaction is completely different, it is somewhat analogous to whipping egg whites: as the time you whip the egg whites increases, you go from liquid egg white, to foamy liquid, to soft, droopy, but “solid” peaks, to hard, shiny, stiff peaks. However, there the similarities end: beating egg whites does not change the molecular structure of eggs, just rearranges the proteins, exposing hydrophobic (water-hating) amino acids and allowing the egg whites to foam into a stiff structure. The process of partial hydrogenation, in addition to adding hydrogen atoms to some carbon double bonds (breaking the double bond and creating a single, saturated bond), tends to change the molecular configuration of any remaining carbon double bonds, leading to trans fatty acids, a creature rarely seen in nature.
So hydrogenation is the process of transforming a liquid oil to a solid or semi-solid fat by breaking carbon double bonds and replacing them with hydrogen-saturated single bonds. Although the reaction is completely different, it is somewhat analogous to whipping egg whites: as the time you whip the egg whites increases, you go from liquid egg white, to foamy liquid, to soft, droopy, but “solid” peaks, to hard, shiny, stiff peaks. However, there the similarities end: beating egg whites does not change the molecular structure of eggs, just rearranges the proteins, exposing hydrophobic (water-hating) amino acids and allowing the egg whites to foam into a stiff structure. The process of partial hydrogenation, in addition to adding hydrogen atoms to some carbon double bonds (breaking the double bond and creating a single, saturated bond), tends to change the molecular configuration of any remaining carbon double bonds, leading to trans fatty acids, a creature rarely seen in nature.
TRANS FATS
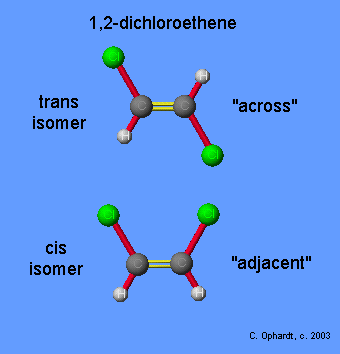 Picture a polyunsaturated fatty acid with 3 carbon double bonds: partial hydrogenation converts 2 of these double bonds to saturated, single bonds. What, if anything, happens to the third double bond? It becomes a trans double bond, yielding a trans fatty acid. To understand how this happens we, once again, turn to our old friend chemistry.
Picture a polyunsaturated fatty acid with 3 carbon double bonds: partial hydrogenation converts 2 of these double bonds to saturated, single bonds. What, if anything, happens to the third double bond? It becomes a trans double bond, yielding a trans fatty acid. To understand how this happens we, once again, turn to our old friend chemistry.
Trans, in this instance, is the opposite of cis, the chemical term for a double bond in which similar atoms are on the same side of the double bond; in trans configuration, the similar atoms are on opposite sides. Because the two molecules have exactly the same constituents (number of carbon, hydrogen atoms, etc.) but different shapes, they are called isomers.
Let’s look again at our example fatty acids, oleic and stearic acid. Both contain 18 carbon atoms and a single carboxyl group; stearic acid contains two extra hydrogen atoms that result in a saturated fatty acid, a straight structure, an increased melting point and the formation of a solid at room temperature. Elaidic acid is another 18 carbon fatty acid, an isomer of oleic acid: oleic acid contains a single carbon double bond in the cis configuration, resulting in a kinked structure, lower melting point and liquid state at room temperature, while elaidic acid contains a single carbon double bond in the trans configuration, resulting in a straighter structure, higher melting point, and semi-solid-to-solid state at room temperature.
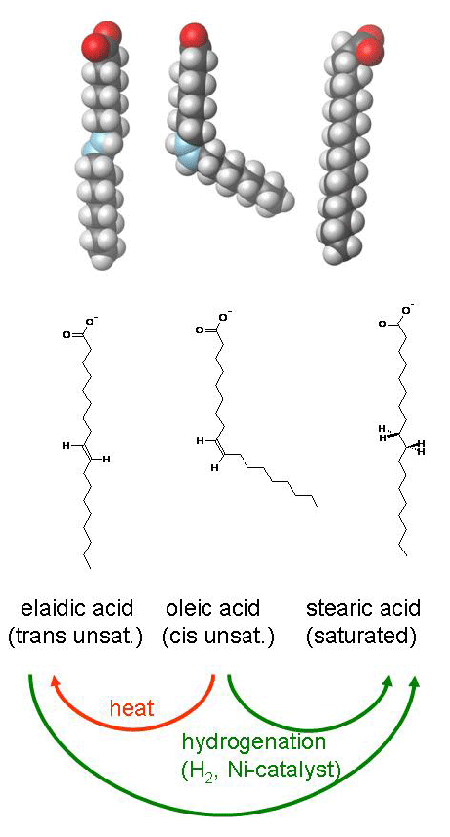 Trans fatty acids are created by partial hydrogenation of unsaturated vegetable oils as a function of the chemical reaction; in the process of bubbling hydrogen gas through the vegetable oil, carbon double bonds are broken and hydrogen atoms are bound to the free carbon sites, as we know. This process happens in three basic steps: 1) the breaking of the carbon double bond; 2) the binding of one hydrogen, and one metal catalyst, to the free carbons, and 3) the binding of the second hydrogen (which replaces the metal catalyst) to produce a saturated, single carbon bond. Step #2 in the reaction, however, is reversible; during this intermediate step the single bound hydrogen is free to spin about the carbon backbone, such that, when the metal catalyst and hydrogen dissociate, the carbon double bond can re-form in either the cis or trans configuration. The trans is the lower energy configuration and is therefore favored in the reaction, so most carbon double bonds that remain in a partially hydrogenated fatty acid will be in the trans configuration, creating the so-called trans fat.
Trans fatty acids are created by partial hydrogenation of unsaturated vegetable oils as a function of the chemical reaction; in the process of bubbling hydrogen gas through the vegetable oil, carbon double bonds are broken and hydrogen atoms are bound to the free carbon sites, as we know. This process happens in three basic steps: 1) the breaking of the carbon double bond; 2) the binding of one hydrogen, and one metal catalyst, to the free carbons, and 3) the binding of the second hydrogen (which replaces the metal catalyst) to produce a saturated, single carbon bond. Step #2 in the reaction, however, is reversible; during this intermediate step the single bound hydrogen is free to spin about the carbon backbone, such that, when the metal catalyst and hydrogen dissociate, the carbon double bond can re-form in either the cis or trans configuration. The trans is the lower energy configuration and is therefore favored in the reaction, so most carbon double bonds that remain in a partially hydrogenated fatty acid will be in the trans configuration, creating the so-called trans fat.
As noted in the diagram above, prolonged heat can also cause cis double bonds to spontaneously rearrange to the trans configuration. This is not to say, however, that sautéing greens in olive oil is going to produce elaidic acid: the temperature, pressure and time required to convert cis fats to trans fats by heat alone is largely impossible in the home kitchen.
USE IN MANUFACTURING
Why partially hydrogenated vegetable oils? Why not simply use the base oil, without processing, or use lard or butter? Well, hydrogenated fats have a number of advantages for food manufacturers. One factor is taste & texture: solid fats (butter, shortening, lard) incorporate into flour better and are responsible for the crisp, airy, or flaky texture of pie crust, cookies, and cakes. In addition, homemade baked goods are usually made with butter: for store-bought cakes “like Mom makes” manufacturers want a similar mouthfeel that liquid oils just don’t deliver.
 Secondly, hydrogenation improves shelf life. Vegetable and seed oils are prone to rancidity because of their very molecular structure: the carbon double bonds are more easily attacked by free radicals, leading to oxygenation and a rancid flavor. Hydrogenating some or all of these bonds improves stability and extends the shelf-life of the products made with hydrogenated fats (I mean, despite what they say, have you ever actually seen a Twinkie rot? I didn’t think so.)
Secondly, hydrogenation improves shelf life. Vegetable and seed oils are prone to rancidity because of their very molecular structure: the carbon double bonds are more easily attacked by free radicals, leading to oxygenation and a rancid flavor. Hydrogenating some or all of these bonds improves stability and extends the shelf-life of the products made with hydrogenated fats (I mean, despite what they say, have you ever actually seen a Twinkie rot? I didn’t think so.)
Thirdly, there is nothing food manufacturers like better than jumping on the latest food trends so that they can make (largely spurious) heatlh claims. Back in the 70’s & 80’s it was all about cholesterol and saturated fat: the USDA and leading health experts were telling us the stuff was poison (and still are), and food manufacturers were happy to fill the need for low saturated-fat, cholesterol-free products.
Lastly, and probably most importantly: partially hydrogenated vegetable oils are cheap. Corn, cotton and soybean are the most heavily subsidized crops in the US, with farm subsidies in the billions paid out in 2005 alone. So it should come as no surprise that corn, cottonseed and soybean oils are dominant in the partially hydrogenated vegetable oil market.
Let’s just think about this for a moment: the most heavily subsidized commodities (our tax dollars at work), some of the most destructive crops on the planet, and the almost all of the crop sources from GMO seed. Even without the reported health risks of trans-fats (which we will delve into in the coming posts), and the fact that, since the January 2006 requirement by the FDA that trans fats should to be listed separately on nutrition labels, trans fat has decreased in our food supply, do we think it’s a good idea that 40% of all supermarket food contains partially hydrogenated vegetable oil?
 Many health sites will tell you that “partially hydrogenated vegetable oil” = “trans fats.” In the past, this was true, but since manufacturers started moving away from trans fats, they have developed processes of hydrogenation that produce less trans fats; not zero trans fats, but less. It’s important to note the the FDA’s labeling rules state that any food containing 0.5 grams or more of trans-fat, per serving, must be labeled; any food containing 0.49 grams and below can be called “0 grams trans fats.” So that bag of chips that you love that has 0 grams of trans fats and the ridiculous 7-chip serving size? Beware. Check ingredient labels; partially hydrogenated vegetable oil still means some amount of trans fats, if less than it once did.
Many health sites will tell you that “partially hydrogenated vegetable oil” = “trans fats.” In the past, this was true, but since manufacturers started moving away from trans fats, they have developed processes of hydrogenation that produce less trans fats; not zero trans fats, but less. It’s important to note the the FDA’s labeling rules state that any food containing 0.5 grams or more of trans-fat, per serving, must be labeled; any food containing 0.49 grams and below can be called “0 grams trans fats.” So that bag of chips that you love that has 0 grams of trans fats and the ridiculous 7-chip serving size? Beware. Check ingredient labels; partially hydrogenated vegetable oil still means some amount of trans fats, if less than it once did.
Another way that food manufacturers can sneak more fat into their products is by adding mono- and diglycerides. Rumor has it that these fatty acids are considered additives by the FDA, since, as we remember from Fat 101, all dietary fats are triglycerides, these mono- and diglycerides are not considered “fats.” However, they are still fatty acid chains; your body digests them in the exact same way that it digests triglycerides (which are broken up into fatty acid components before being transported, stored or used by the body). As far as I can tell, since mono- and diglycerides are not considered “fat,” there is no regulation governing the reporting of these fatty acids: they could be fully saturated, unsaturated, trans fatty acids, or a mix, without being reported on the label. (Note: I’m unable to find confirmation of this ruling on the FDA’s (byzantine) website. There are some vague statements regarding “limited use” of ingredient additives, but nothing I see that is concrete). Just be aware that anything that looks like a fat: any type of oil, generally anything labeled “acid” appearing in the first few ingredients (unless you are eating Sour Patch Kids, that is), anything with “glyceride” or “lipid” in the label, well it probably is a fat. And probably not one found in nature.
 Also be aware that not all trans fats are created equal. While trans fats do exist, in small amounts, in nature, they are not formed by animals or plants. Milk or meat products from ruminant animals (cows, sheep, goats) can contain 2-5% of natural trans-fat, produced by the bacteria that exist (and aid in digestion) in the rumen. However, the largest source (about 80%, according to the USDA) of trans fats in the American diet is by far processed foods, and there is some indication that natural trans fats, which are metabolized in the body to conjugated linoleic acid (CLA, reputed to have anti-cancer effects), do not pose the health risks associated with manufactured trans fats. Which leads us to our next topic: essential fatty acids.
Also be aware that not all trans fats are created equal. While trans fats do exist, in small amounts, in nature, they are not formed by animals or plants. Milk or meat products from ruminant animals (cows, sheep, goats) can contain 2-5% of natural trans-fat, produced by the bacteria that exist (and aid in digestion) in the rumen. However, the largest source (about 80%, according to the USDA) of trans fats in the American diet is by far processed foods, and there is some indication that natural trans fats, which are metabolized in the body to conjugated linoleic acid (CLA, reputed to have anti-cancer effects), do not pose the health risks associated with manufactured trans fats. Which leads us to our next topic: essential fatty acids.
ESSENTIAL FATTY ACIDS
The human body can maufacture most of the fatty acids that it needs to perform bodily funtions from the assortment of dietary fats that we consume. There are two fatty acids, termed the essential fatty acids, that the body can not manufacture: alpha-linolenic acid (ALA) and linoleic acid (LA). Alpha-linolenic acid is also known as an omega-3 fatty acid, so named because the first carbon double bond is three carbons in from the non-carboxyl, or omega, end of the fatty acid chain (versus the first carbon at the carboxyl end, which is called the alpha carbon).
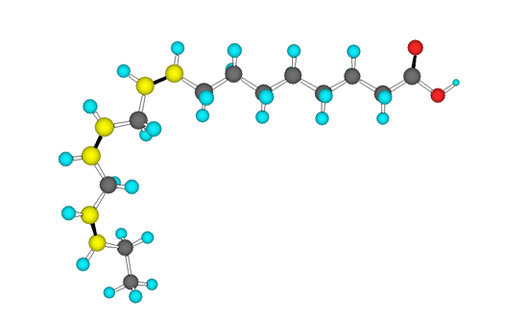 Alpha-linolenic acid is known as a base omega-3, from which other omega-3 fatty acids, including DHA and EPA, can be synthesized. Omega-3 fatty acids confer a number of reputed health benefits, cheif among them prevention of heart diesease. The main dietary sources of omega-3 fatty acids, including the essential fatty acid ALA, are found in fish (especially oily fish), flax and hemp seeds, walnuts, and meat, milk and eggs from pastured or wild animals. Ligonberry, purslane and sea buckthorn are also food sources high in ALA.
Alpha-linolenic acid is known as a base omega-3, from which other omega-3 fatty acids, including DHA and EPA, can be synthesized. Omega-3 fatty acids confer a number of reputed health benefits, cheif among them prevention of heart diesease. The main dietary sources of omega-3 fatty acids, including the essential fatty acid ALA, are found in fish (especially oily fish), flax and hemp seeds, walnuts, and meat, milk and eggs from pastured or wild animals. Ligonberry, purslane and sea buckthorn are also food sources high in ALA.
Similar to the nomenclature for ALA, an omega-3 fatty acid, linolenic acid, or LA, is known as an omega-6 fatty acid, due to the position of the first carbon double bond at the sixth carbon from the omega end of the chain.
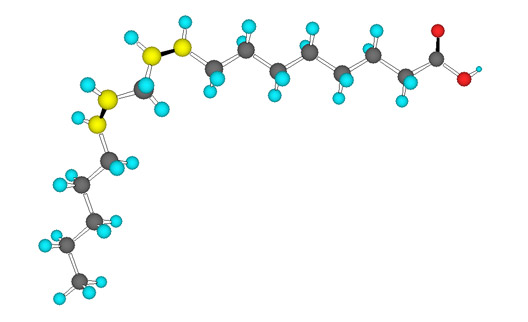 Linolenic acid is abundant in many vegetable oils, including corn, soybean and cottonseed, and therefore, the American diet includes plentiful omega-6 fatty acids. Although humans clearly need omega-6 fatty acids, especially the essential fatty acid LA, research suggests that the ratio of omega-6 to omega-3 fatty acids is important for overall good health (the current US ratio of dietary omega-6:omega-3 is approximately 16:1. Evidence suggests that humans evolved eating a ratio of 1:1 and ratios ranging from 5: 1 to 2.5:1 have shown beneficial impact on cancer, heart disease and various inflammatory diseases). So, in order to improve this ratio, the Dietary Guidelines tell us to decimate fish populations (“all Americans should increase their intake of fish from 3 and 1/2 oz to 8 oz per week”) rather than telling us to eat less omega-6 fatty acids, found in the vegetable oils prevalent in processed foods. We’ll delve further into this recommendation in the coming weeks.
Linolenic acid is abundant in many vegetable oils, including corn, soybean and cottonseed, and therefore, the American diet includes plentiful omega-6 fatty acids. Although humans clearly need omega-6 fatty acids, especially the essential fatty acid LA, research suggests that the ratio of omega-6 to omega-3 fatty acids is important for overall good health (the current US ratio of dietary omega-6:omega-3 is approximately 16:1. Evidence suggests that humans evolved eating a ratio of 1:1 and ratios ranging from 5: 1 to 2.5:1 have shown beneficial impact on cancer, heart disease and various inflammatory diseases). So, in order to improve this ratio, the Dietary Guidelines tell us to decimate fish populations (“all Americans should increase their intake of fish from 3 and 1/2 oz to 8 oz per week”) rather than telling us to eat less omega-6 fatty acids, found in the vegetable oils prevalent in processed foods. We’ll delve further into this recommendation in the coming weeks.
NEXT WEEK
This has been a lot to take in, and I’ve decided that cholesterol needs its very own post, so stay tuned: next week we’ll discuss Cholesterol: The Good, The Bad & The Ugly. As always, please feel free to ask questions or provide any feedback: it is my hope that these posts are informative without being painful, and maybe even a little bit fun.
Sources: Lawry’s garlic powder image from befoodsmart.com. Saturated, mono- and polyunsaturated molecular models from Wikipedia Commons, linked directly in Fat 101. Hydrogenation of oleic acid diagram from here. Marvin Gaye album cover image from Wikipedia. Vintage Crisco ad here. Cis and trans model from Elmhurst. Trans fat model diagram from UMass Amherst. I Want To Believe poster here. Zero grams trans fat label from Wellsphere. Cow picture from EcoFriendly Daily. Omega-3 and omega-6 molecular models from omega-3 learning by the University of Connecticut. Cholesterol image from Discovery Health. Happy Pig from amyzyng via Flickr Creative Commons. All learning sources are linked directly in the text.


I haven’t even finished reading yet but I just want you to know that you absolutely KILL me … (a little soft lighting, a little Marvin Gaye)! I’m going to pee myself, I just know it!!
I’ve been MIA from blogs lately and just got all caught up on this series, which is such a fabulous idea! I’m so glad you’re doing it. I will say though, at times you’re making my head hurt. Although you’re explaining the chemistry really well, that was a subject I was never truly fond of and I’m struggling to remember back several (7?) years so I’m not so lost. I’m just glad ESF decided Wildlife Science majors didn’t need to know Organic Chemistry!
Can’t wait to read more!
Ha! I took Organic Chem in the summer of ’88 – it was quite some time ago. Despite the fact that chemistry is inherent in my work, it was never my favorite; I’m a biologist through and through. But chemistry and biology are hopelessly intertwined, so trust me, all of this chemistry is leading somewhere! Hopefully to a better undertstanding of the biology, or what fats do in our bodies and how the fats we eat impact our health. Or at least – that’s the plan.
p.s. I can tell you that, at times, this post made *my* head hurt. 🙂
Pingback: {weekend reading} NATIONAL EDITION « FROM SCRATCH club
wow! i commend you, i really do. and from this right-brained, left-handed cat i can tell you that you make learning fun. i proclaimed my head was swimming from 101 (drowning was probably more accurate) i am treading for 102, and i have a feeling i’ll be catching waves by the time you’re through.
thank you, this is really important work.
I love that pig! And you are a genius!
Wow. You have an incredible ability to take a very complex topic and explain it in a very concise and clear way. Though I teach high school science (and just finished teaching a biochem unit ) I never took O Chem myself. Thanks for the education!
High praise indeed! Thanks, Karen!