 Fat. Saturated fat. Unsaturated fat. Monounsaturated, polyunsaturated, partially hydrogenated, trans fat. Lipids, fatty acids, triglycerides, cholesterol, omega-3, omega-6. No wonder we’re confused! What does it all mean?
Fat. Saturated fat. Unsaturated fat. Monounsaturated, polyunsaturated, partially hydrogenated, trans fat. Lipids, fatty acids, triglycerides, cholesterol, omega-3, omega-6. No wonder we’re confused! What does it all mean?
From the OED online:
Pronunciation:/fat, / noun 1) a natural oily or greasy substance occurring in animal bodies, especially when deposited as a layer under the skin or around certain organs, 2) a fatty substance made from animal or plant products, used in cooking 3) any of a group of natural esters of glycerol and various fatty acids, which are solid at room temperature and are the main constituents of animal and vegetable fat.
So, you know that I am taking on the USDA’s Dietary Guidelines for Americans, separating the wheat from the chaff, the good information from the techno-babble, and trying to distill all it into a form that non-government, non-scientist humans can understand. One of the key recommendations throughout that document regards fat: they want us to eat less of it. They site clinical studies that implicate dietary fat in a host of disease states: heart disease, atherosclerosis, diabetes, cancer. But are they looking at all the appropriate studies, or picking & choosing based upon selective criteria? Are they biased by the weight of scientific evidence from years past, or are they viewing recent studies with open eyes? And are the studies themselves asking the right questions?
 It’s a complex topic, and I’ll tell you right now that there are many conflicting opinions and few clear-cut answers. But I realized that before we can delve into the heart of the matter, we need to understand exactly what fat is: what all the terms mean, how they interrelate and how the scientific terms describe what ends up on our plate. Now, to learn exactly what fat is, we’re going to need to resort to – hang onto your hats – chemistry. Because chemistry is life, people (up until the point where physics is life) and at some point, we need to grasp a little bit of chemistry if we’re going to understand life. Think of it this way: chemistry is form, while biology is function. Since form follows function, understanding the chemistry can make the biology obvious, or at the very least, more intuitive. And I promise it won’t hurt. I’ll give you a lollipop after. Are you with me? OK, let’s go!
It’s a complex topic, and I’ll tell you right now that there are many conflicting opinions and few clear-cut answers. But I realized that before we can delve into the heart of the matter, we need to understand exactly what fat is: what all the terms mean, how they interrelate and how the scientific terms describe what ends up on our plate. Now, to learn exactly what fat is, we’re going to need to resort to – hang onto your hats – chemistry. Because chemistry is life, people (up until the point where physics is life) and at some point, we need to grasp a little bit of chemistry if we’re going to understand life. Think of it this way: chemistry is form, while biology is function. Since form follows function, understanding the chemistry can make the biology obvious, or at the very least, more intuitive. And I promise it won’t hurt. I’ll give you a lollipop after. Are you with me? OK, let’s go!
According to my college Biology textbook [4th ed, 1986(!)], lipids are a general category of compounds that do not dissolve in water, but readily dissolve in solvents such as acetone (think nail polish remover – by far the easiest way to get peanut butter out of a child’s hair). Fats fall into the lipid category (along with cholesterol and other steroids, phospholipids, soaps & detergents, and some vitamins). Hence all fats are lipids, but not all lipids are fats. With me so far?
 Picture this: you take a blueberry; you stick three toothpicks in it. This is your basic representation of a fat molecule. The toothpicks are your fatty acids: long, straight chains of carbon atoms, linked hand-in-hand like a pre-school class on a field trip. The blueberry is your glycerol, the compound that packages the fatty acids into groups of three. Dietary fat molecules are composed of three fatty acids all bound to a glycerol, hence the name triglyceride (fat molecules can also be mono- or diglycerides, but all dietary fats are triglycerides).
Picture this: you take a blueberry; you stick three toothpicks in it. This is your basic representation of a fat molecule. The toothpicks are your fatty acids: long, straight chains of carbon atoms, linked hand-in-hand like a pre-school class on a field trip. The blueberry is your glycerol, the compound that packages the fatty acids into groups of three. Dietary fat molecules are composed of three fatty acids all bound to a glycerol, hence the name triglyceride (fat molecules can also be mono- or diglycerides, but all dietary fats are triglycerides).
Fatty acids are the long chains of linked carbon atoms (the toothpicks, or chain of kids holding hands, represented by the grey strands at left). The white balls are hydrogen atoms, bound to carbon (picture every child in the pre-school chain wearing a white hat and white shoes). The glycerol (blueberry) is in red. So, to review: a fat molecule consists of three fatty acids, each a chain of carbon atoms bound to hydrogen atoms, all held together at one end by a glycerol. (For the chemistry-inclined, this page shows the C-H bond structures of various fatty acids.)
So, now you have a triglyceride. But, since all fats have a glycerol in common, the fatty acids are where the action is: differences in fatty acid structure relate to differences in performance, and impact on human health (form follows function again). Remember those white balls? They represent the hydrogen atoms, bound to the carbon chain that is the backbone of the fatty acid. A fatty acid in which every ‘free’ carbon binding site (all the kids’ heads and feet; the hands are already ‘bound’ to each other in the line) is bound to a hydrogen is called saturated (every kid is wearing a white hat and white shoes); the carbon chain is saturated with hydrogen atoms. Likewise, a carbon chain that does not contain a hydrogen at every potential carbon bonding site is called unsaturated (some of the kids, with wild abandon, took off their hats and left them in their lockers).
Those ‘free’ carbon sites, unbound to a hydrogen, will form a bond with a neighboring carbon (the notation being a double bond, since the carbons are already bound once in the backbone; i.e. the kids are already holding hands; when the teacher knocks their heads together for not wearing their hats (hey, it could happen) they are “bound” twice – at the hand and at the head). This double bond produces a kink in the backbone (picture the kids trying to walk in a straight line if their heads are stuck together), hence unsaturated fats have a bent or kinked shape while saturated fats lay flat. A monounsaturated fat contains one carbon double bond (one kink); a polyunsaturated fat contains two or more double bonds (seriously kinky).
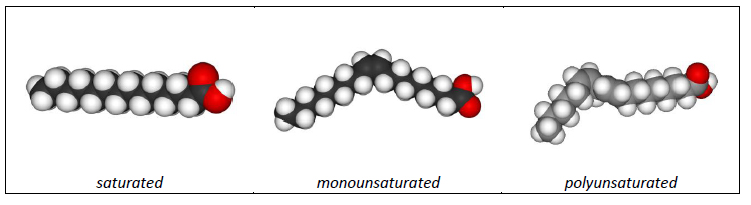 Things makes sense when you look at the physical structures of a single fatty acid (above): saturated fats (butter, lard, bacon grease, etc.) are generally solid at room temperature, whereas unsaturated fats (olive and most other vegetable oils) are liquid at room temperature. Saturated fats, with all those nice hydrogen bonds in a row, have straight fatty acids, which allow them to lie flat and plate against each other to form a more compact overall product. This compact structure relates to strong intra-molecular bonds, meaning that butter has a higher melting point (more energy, in the form of heat, is required to break the bonds and melt the butter). Unsaturated fats, because of the kinks in their fatty acid structure caused by the missing hydrogen and resulting carbon double bonds, are unable to lie flat; therefore they form a slightly less dense, liquid product at room temperature (with weaker intra-molecular bond strength) and a lower melting point. To review: saturated fats have straight fatty acids, complete hydrogen bonding, and are solid at room temperature. Unsaturated fats have bent or kinked fatty acids, one (mono) or more (poly) carbon double bonds, and are liquid at room temperature.
Things makes sense when you look at the physical structures of a single fatty acid (above): saturated fats (butter, lard, bacon grease, etc.) are generally solid at room temperature, whereas unsaturated fats (olive and most other vegetable oils) are liquid at room temperature. Saturated fats, with all those nice hydrogen bonds in a row, have straight fatty acids, which allow them to lie flat and plate against each other to form a more compact overall product. This compact structure relates to strong intra-molecular bonds, meaning that butter has a higher melting point (more energy, in the form of heat, is required to break the bonds and melt the butter). Unsaturated fats, because of the kinks in their fatty acid structure caused by the missing hydrogen and resulting carbon double bonds, are unable to lie flat; therefore they form a slightly less dense, liquid product at room temperature (with weaker intra-molecular bond strength) and a lower melting point. To review: saturated fats have straight fatty acids, complete hydrogen bonding, and are solid at room temperature. Unsaturated fats have bent or kinked fatty acids, one (mono) or more (poly) carbon double bonds, and are liquid at room temperature.
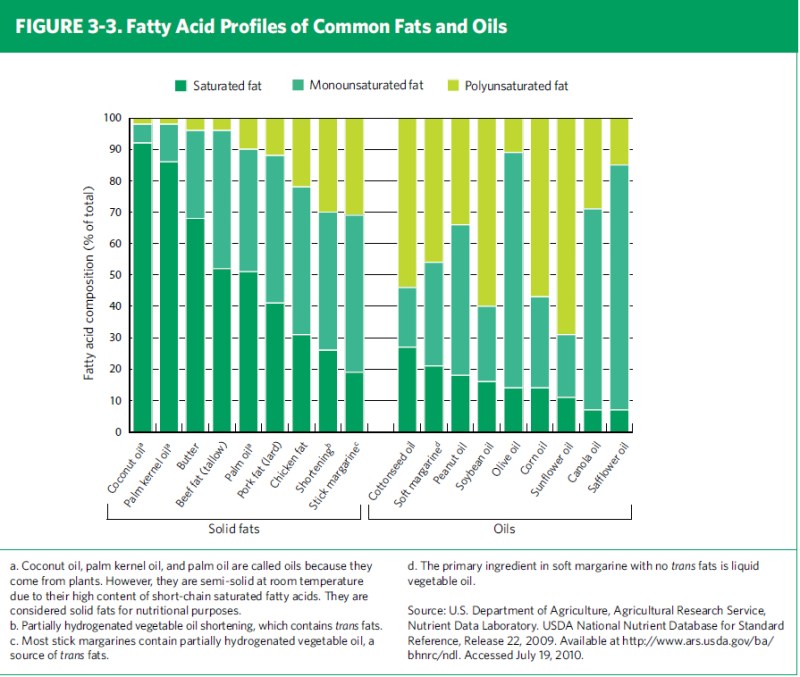 Now, while we talk about butter as a saturated fat, and olive oil as an unsaturated fat, in reality, all fats contain a mixture of saturated, mono- and polyunsaturated fats (in fact, triglycerides can contain a mix of saturated and unsaturated fatty acids); solid fats contain more saturated fats and less unsaturated fats, while liquid fats are the opposite. The type of fatty acid that represents the largest proportion of the particular fat tends to be how that fat is labeled, for example, olive oil is often referred to as a monounsaturated fat, when it reality, it contains all three types of fatty acids. It’s the largest proportion of fatty acid type that is considered representative of a fat or oil.
Now, while we talk about butter as a saturated fat, and olive oil as an unsaturated fat, in reality, all fats contain a mixture of saturated, mono- and polyunsaturated fats (in fact, triglycerides can contain a mix of saturated and unsaturated fatty acids); solid fats contain more saturated fats and less unsaturated fats, while liquid fats are the opposite. The type of fatty acid that represents the largest proportion of the particular fat tends to be how that fat is labeled, for example, olive oil is often referred to as a monounsaturated fat, when it reality, it contains all three types of fatty acids. It’s the largest proportion of fatty acid type that is considered representative of a fat or oil.
Well, kids, I think that’s enough for today. Join us in the next class when we talk about trans fats, hydrogenation and the demon cholesterol, which isn’t actually a fat at all, but a steroid (no, not the Barry Bonds kind). As homework, please give a holla below if you have any feedback on today’s class: Was it helpful? Boring? Too complicated? Too simplistic? Too long? Too short? Are you all jazzed up to learn more about fat, or are you just waiting for me to make more jam? I’d be happy to hear your thoughts.
Sources: Wikipedia does a nice job on fat, with lots of citations and good sub-pages on the various forms. A veritable geek-fest on this lipids page from Michigan State. This page from UC Clermont has some nice diagrams and clear descriptions of various types of lipids. Molecular structure diagrams were prepared by Ben Mills, used via Wikipedia Commons. Fatty acid profile plot from the Dietary Guidelines 2010.
Disclaimer: Organic chemistry is not my forté and it’s been quite some time since I’ve thought about these concepts. I’ve culled this information from various textbooks and (hopefully) reputable online sources; however, if you are a chemist, teacher, student or enthusiast and you notice something above that is misleading, or flat-out wrong, I do hope you shout out it below.

First of all, I love you. In the way that I also love Alton Brown, not in the much different way that I love my husband.
Second of all, be a teacher.
That is all.
🙂
Aw, thanks! I always thought I would make a good biology teacher, but I’m saving it for when I’m 90, so I can toss around terms like “whippersnapper” and “poppycock.” It’ll be a grand old time. 🙂
I’m loving it so far! In fact, I was a tad disappointed by the cliff hangar, because I was so into it (picturing all those little children walking hand-in-hand on a field trip!) Can’t wait for the next episode 🙂
Thanks, Nancy, that’s helpful feedback. I could’ve kept going & going, but then, I’m a geek and I love this stuff. I’d figured I’d stop before I made all of your heads explode…. 🙂
Stumbled upon your site thru facebook and a friend on there. Am fighting off a virus so am gonna have to come back later to read your post because my head is already pounding and it would take very little to make it explode. I am hoping I can make heads to tails what you have shared so that I can start making better choices about what to eat and not to eat. Already figured out that HFCS is a bad thing. And GM stuff isn’t real food either. Fats are a curious thing because I know we need them in some forms….. anyway… off to lay down again.. book marked your page. Thanks for letting me ramble….
Thank you so much for this! It’s exactly what I need/want. Please continue I want to know the science and have a real explanation for all this dietary recommendation craziness!
hey kaela,
i must admit my head is swimming a bit (def did better in art than science in school!) but i am following along and i commend you for attempting to dig into the dietary guidelines – mess that it is.
Pingback: {weekend reading} NATIONAL EDITION « FROM SCRATCH club
I love you Kaela. I bought Jennifer McLagan’s cookbook called ‘Fat: an appreciation of a misunderstood ingredient, with recipes’ last year when I was trying to combat my editor’s chick-magazine understanding of healthy cooking (low-fat, butter-less, meh, blech). I am so appreciative of this wealth of info you’ve compiled here (and your blueberry model to boot!).
xxoo
Thanks, everyone. Working on the next installment as we speak! Trans fats, hydrogenated fats and cholesterol, oh my!
thank you so much for delving into this huge topic. i am a bit fan of learning more about all our foods, and i think fat especially is a topic that people are misguided about. during my cooking classes there are always questions and misconceptions about what/how much/why certain fats are healthy or not. i would love to repost this on my blog… is that ok with you? i am bakerymanis.wordpress.com- growing, baking and eating on the island :0
and, i have a book recommendation for you! twinkie, deconstructed: my journey to discover how the ingredients found in processed foods are grown, mined and manipulated in what america eats by Steve Ettlinger. it’s a funny, informative, and even handed look at how twinkies (and other processed and not-so-processed foods) are made.